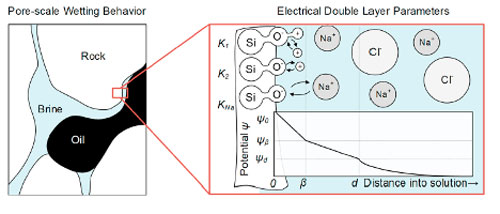
Scientists carry out electrochemical studies on physicochemical systems for a variety of reasons including obtaining thermodynamic data, understanding the kinetics of system evolution of degradation, or analyzing a solution for trace amounts of chemical compounds. There are also investigations in which the system's electrochemical properties themselves are of primary interest, for example the design of new power generation systems (e.g., fuel cells and batteries) and synthesis of new materials such as metal alloys or oxide films.
The Electrochemical Technologies Program at the EMS Energy Institute strives to be at the forefront of fundamental and applied research in a variety of electrochemical and materials science technologies. It aims to promote and facilitate the use of electrochemical probes and systems in areas of science and technology important for society, in particular fuel cells and electrolysis, materials for deep well drilling, hydrothermal synthesis of new materials, and electrophoresis of nanoparticles. The specific expertise of this program is related to interfacial electrochemistry and corrosion in extreme environments (e.g., high temperature and pressure and high concentration).